- Home
- About the Journal
- Peer Review
- Editorial Board
- For Authors
- Reviewer Recognition
- Archive
- Contact
- Impressum
- EWG e.V.
Cite as: Archiv EuroMedica. 2026. 16; 1. DOI 10.35630/2026/16/Iss.1.004
Hyperthyroidism and anxiety disorders are highly prevalent conditions that frequently coexist and present with substantial clinical overlap. Their comorbidity is associated with impaired functioning, reduced quality of life, and diagnostic challenges that may lead to delayed or incomplete treatment.
The aim of this narrative review was to systematize current evidence on the relationship between hyperthyroidism and anxiety disorders, with particular emphasis on shared pathophysiological mechanisms, clinical symptom overlap, impact on quality of life, and implications for interdisciplinary management.
A narrative review of the literature was conducted using publications identified in PubMed, PubMed Central, and Google Scholar. The search was performed between October 15 and November 10, 2025, without a strict time restriction. A total of 44 relevant sources, including narrative and systematic reviews, meta analyses, observational clinical studies, and experimental preclinical studies, were included and synthesized descriptively.
The analyzed literature demonstrates a complex and bidirectional relationship between hyperthyroidism and anxiety disorders. Key findings indicate significant neuroendocrine dysregulation involving interactions between the hypothalamic pituitary thyroid and hypothalamic pituitary adrenal axes, alterations in neurotransmitter systems, autoimmune mediated processes, and emerging contributions of the gut brain axis. Clinically, substantial symptom overlap frequently complicates differential diagnosis, and anxiety symptoms often persist despite restoration of euthyroidism, leading to sustained impairment in quality of life.
Hyperthyroidism represents a clinically relevant and often underrecognized contributor to anxiety related symptomatology. Normalization of thyroid hormone levels alone is frequently insufficient to resolve psychiatric symptoms, underscoring the need for integrated interdisciplinary management. Future research should focus on longitudinal and interventional studies to clarify underlying mechanisms and to evaluate targeted psychiatric and endocrine treatment strategies aimed at improving long term functional outcomes and quality of life.
Keywords: hyperthyroidism, Graves disease, anxiety disorders, quality of life, neuroendocrine mechanisms, HPT axis, HPA axis.
Hyperthyroidism (HT) is an endocrine condition characterized by the excess synthesis and secretion of thyroid hormones, namely triiodothyronine (T3) and/or free thyroxine (fT4), resulting in circulating levels that exceed physiological tissue demand and subsequently lead to a spectrum of well-characterized symptoms of varying severity [1]. The overall prevalence of hyperthyroidism is estimated at 1.2% in the general population, with the lifetime risk assessed at 3% in women and 0.5% in men. The most common etiology of hyperthyroidism is Graves' Disease (GD), which accounts for 60–80% of all cases. GD primarily affects individuals between the ages of 20 and 50 years but can manifest at any age [2]. Other significant causes of thyrotoxicosis include, but are not limited to, toxic nodules (solitary or multinodular goiter) and the thyrotoxic phase of thyroiditis [1].
Anxiety disorders represent a diverse group of highly prevalent mental health conditions, defined by core features such as excessive worry, social apprehension and performance fears, unexpected or context-dependent panic attacks, pervasive anticipatory anxiety, and subsequent avoidance behaviors. These disorders constitute a major public health concern, with estimates indicating a lifetime prevalence reaching approximately 34% in the United States. Within this group, the most frequently diagnosed conditions include social anxiety disorder (SAD), reported to have a lifetime prevalence of 13%, generalized anxiety disorder (GAD), affecting about 6.2% of the population, and panic disorder, with a lifetime prevalence of 5.2% [3].
Hyperthyroidism is characterized by a high prevalence of anxiety-related symptoms, including palpitations, internal restlessness, psychomotor agitation, and fine motor tremor. These manifestations frequently lead to an initial misdiagnosis of primary psychiatric disorders, which invariably results in delayed detection and treatment of the underlying endocrine dysfunction. Furthermore, formal psychiatric consultations for patients with confirmed hyperthyroidism are often insufficient or implemented at a late stage, only after the psychiatric symptoms have become entrenched or fully developed [2].
The aim of this narrative review is to systematize and analytically synthesize current evidence on the relationship between hyperthyroidism and anxiety disorders, with a focus on shared pathophysiological mechanisms, clinical symptom overlap, the impact of psychiatric comorbidity on quality of life, and existing therapeutic approaches.
This work represents a narrative literature review. The analysis is based on published scientific sources included in the References list of this article.
The literature search was conducted between October 15 and November 10, 2025, using the electronic databases PubMed, PubMed Central, and Google Scholar. The search was performed without a strictly predefined time restriction. Priority was given to more recent publications, while earlier sources were included when they were directly relevant to the topic and had fundamental significance.
During the search, key terms reflecting the endocrine and psychiatric aspects of the topic were used, including hyperthyroidism, Graves disease, thyrotoxicosis, anxiety disorders, psychiatric symptoms, and quality of life. Additional pathophysiological concepts, including the HPT and HPA axes and the gut brain axis, were considered at the stage of analysis and interpretation of the selected sources.
A total of 44 sources were included in the final analysis. The literature set comprises narrative reviews, systematic reviews and meta analyses, original observational clinical studies, experimental preclinical studies, as well as selected fundamental publications relevant to the pathophysiological, clinical, and psychiatric aspects of thyroid diseases.
The analysis excluded single case reports, case series without an analytical component, editorials, commentaries, letters to the editor, conference materials, abstracts, non peer reviewed publications, as well as studies focusing exclusively on hypothyroidism or psychiatric disorders without assessment of thyroid function. Publications that did not contain data or discussion relevant to anxiety symptoms or anxiety disorders in the context of hyperthyroidism or autoimmune thyroid disease were also excluded.
Definitions of hyperthyroidism and anxiety disorders were not unified and were used as presented in the respective primary sources. Methods for assessing anxiety symptoms, psychiatric manifestations, and endocrine parameters varied across studies.
Data extraction was performed descriptively and included the type of publication, the studied population or experimental model, the clinical and pathophysiological aspects addressed, and the main findings relevant to the relationship between hyperthyroidism and anxiety disorders. Results were synthesized descriptively without quantitative pooling of data and without formal assessment of methodological quality of the included studies.
The comprehensive analysis of the selected literature reveals a multifaceted and bidirectional relationship between hyperthyroidism and anxiety disorders. The review identified four primary domains of interaction: shared pathophysiological mechanisms, clinical overlap leading to diagnostic challenges, the impact on quality of life (QoL), and therapeutic implications. Biologically, the literature consistently points to neuroendocrine dysregulation involving the interaction between the hypothalamic-pituitary-thyroid (HPT) and hypothalamic-pituitary-adrenal (HPA) axes, as well as alterations in monoaminergic neurotransmission [5, 9]. More recent studies highlight the emerging role of the gut-brain axis and microbiota dysbiosis as significant contributors to the psychiatric manifestations of autoimmune thyroid diseases [13, 15]. Clinically, the data indicates a high rate of phenomenological overlap, where somatic symptoms of thyrotoxicosis mimic primary anxiety disorders, frequently resulting in misdiagnosis or delayed treatment [2, 19]. Furthermore, evidence suggests that achieving euthyroidism does not guarantee the remission of psychiatric symptoms, which continue to significantly impair patients' functional status and QoL [2, 20]. The following subsections present a detailed synthesis of these findings.
The most common etiology of hyperthyroidism, Graves’ disease (GD), is an autoimmune disorder resulting from the activation of thyroid-stimulating hormone (TSH) receptors by specific thyroid-stimulating antibodies. Elevated circulating thyroid hormone levels enhance catecholaminergic activity by promoting an increased density of beta-adrenergic receptors on the cellular surface [4]. This neuroendocrine effect leads to some of the most prominent somatic symptoms, such as palpitations, diaphoresis (sweating), and fine tremor, all of which are frequently observed in anxiety disorders. These manifestations typically dominate the initial phase of thyrotoxicosis. In contrast, chronic hyperthyroidism can lead to persistent emotional instability and a heightened propensity for developing mood disturbances.
Psychiatric symptoms observed during hyperthyroidism are postulated to stem, in part, from an imbalance of crucial neurotransmitters such as serotonin, noradrenaline, and dopamine, a phenomenon evidenced in various animal models [5]. Thyroid hormones (thyronines) and their metabolic derivatives (including thyronamines and thyroacetic acids) are concentrated within brain tissue. Although the substantial accumulation of thyroid hormones in nerve terminals suggests a potential role in synaptic function, little is currently understood regarding the potential release of thyroid hormones at the synaptic clefts [6]. Specific binding sites for thyroid hormones exist within synaptosomes, including the integrin αvβ3 receptor and other neurotransmitter receptors, where they exert allosteric effects on major systems, including GABAergic, catecholaminergic, glutamatergic, serotonergic, and cholinergic pathways. While some literature suggests that thyroid hormones and their derivatives could function as neurotransmitters, they fulfill most, but not all, established defining criteria [6]. The pathophysiology of thyroid hormone-related changes in cerebral tissue is complex, involving alterations in neurotransmission, changes in gene expression in neuronal and glial cells, disruption of the blood-brain barrier (BBB), and, occasionally, cerebral inflammatory disease, particularly within the context of autoimmune conditions like Graves’ disease. Since the number of known TH-responsive genes in adult cerebral tissue appears limited, the overall effect of TH may be predominantly post-transcriptional or initiated at non-nuclear receptor sites. Specifically, thyroid hormones increase both the quantity and responsiveness of beta-adrenergic receptors in synaptosomal membranes and cerebral microvessels [7]. Thyroid hormones also exert significant influence on other endocrine systems, notably the hypothalamic-pituitary-adrenal (HPA) axis, creating intricate interactions among various neuroendocrine pathways [8]. Murine models of induced hyperthyroidism have demonstrated elevated levels of serotonin and noradrenaline in brain samples, which are neurotransmitters commonly implicated in anxiety disorders [9].
The complete pathophysiology of primary anxiety disorders remains under investigation, though neurobiological, genetic, and environmental factors are recognized contributors to their onset. One of the most critical neurobiological factors is thought to be the altered neurotransmission in specific brain regions. These changes include heightened activity in emotional processing areas, possibly due to weakened inhibitory signaling mediated by γ-aminobutyric acid (GABA), alongside reduced serotonergic neurotransmission [10]. Studies have shown that compounds blocking the serotonin transporter (SERT) and acting as antagonists at 5-HT2A, 5-HT2C, and 5-HT7 receptors-thereby increasing levels of serotonin (5-HT), norepinephrine (NE), dopamine (DE), and glutamate in the prefrontal cortex-effectively reduced stress-related behaviors in social interaction tests [10]. Consequently, medications that inhibit the synaptic reuptake of serotonin (SSRIs) and serotonin and norepinephrine (SNRIs) are considered the first-line pharmacotherapy for most anxiety disorders [3].
The persistent activation of the HPA axis, often triggered by stressful experiences during developmental stages, is also hypothesized to increase the risk for anxiety disorders [8, 10]. The regulatory role of the HPA axis in anxiety has been highlighted in multiple studies [10]. Furthermore, clinical investigations have noted a tendency toward lower TSH concentrations and higher free thyroxine fT4 levels in patients presenting with primary anxiety disorders [11]. Continued research into the precise interactions between the hypothalamic-pituitary-thyroid (HPT) axis and the HPA axis is necessary, as these systems appear to hold a significant role in the shared pathomechanism of both condition groups.
Genetic factors suspected of contributing to the development of anxiety disorders primarily include variants of the THEM132D gene, whose products regulate the HPA axis and monoaminergic signaling, as well as the intragenic region of NTRK2 (encoding the tropomyosin receptor kinase B (TrkB) for brain-derived neurotrophic factor (BDNF)), which is also implicated as a key modulator [10]. Additionally, fluctuations in levels of neurotrophins, such as nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF)-molecules vital for synaptic plasticity, neuronal function, and cell survival-are observed in Graves’ disease and mirror those found in primary psychiatric disorders, such as anxiety, where these processes are disrupted [12].
The gut-brain axis represents an emerging target for research into psychiatric symptoms linked to autoimmune conditions [13]. Graves’ disease (GD) is consistently associated with an altered gut microbiome (dysbiosis) when compared to healthy controls [14, 15]. This microbial imbalance, potentially contributing to systemic inflammation, is hypothesized to modulate neurotransmitter pathways and disrupt the blood-brain barrier, thereby leading to or exacerbating anxiety and mood symptoms, presenting a crucial novel pathomechanism for psychiatric comorbidity [13].
In recent years, the inflammatory basis of psychiatric disorders has gained increasing attention. Several studies have explored the role of neuroinflammation and the Nitric Oxide (NO) pathway in conditions such as juvenile social stress and anxiety-like behavior related to obesity-induced neuroinflammation [16, 17]. Nevertheless, future targeted research is still needed to specifically investigate the role of inflammatory mediators inherent to Graves’ disease (as an autoimmune inflammatory condition) and their contribution to the pathogenesis of co-occurring psychiatric disorders.
The interaction between thyroid hormone excess, neuroendocrine stress regulation, immune mediated processes, and microbiota related mechanisms is discussed below and visually summarized to illustrate their convergence in the development of anxiety related symptoms in hyperthyroidism, as shown in Figure 1.
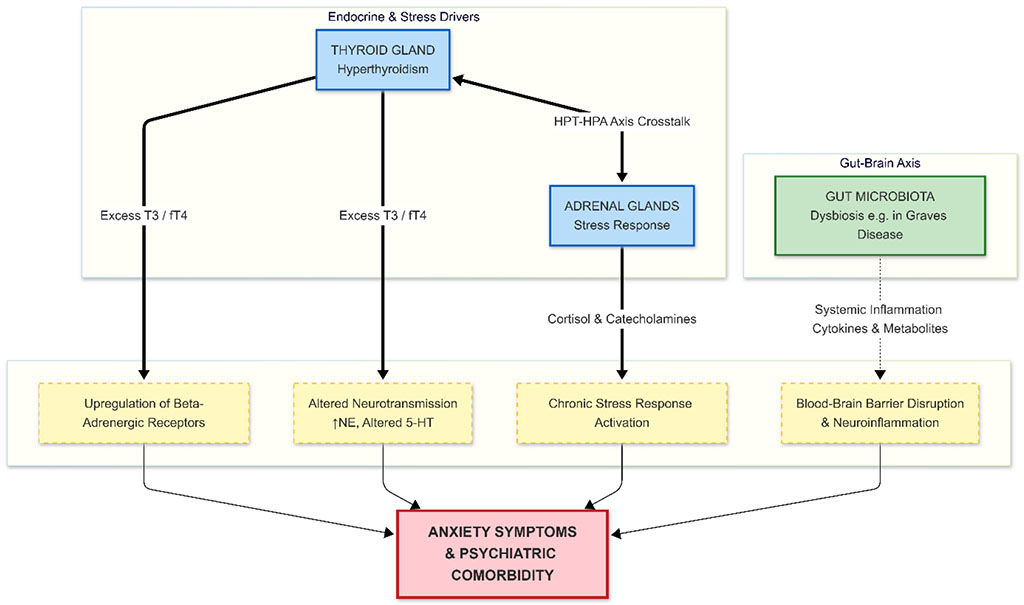
Figure 1. Schematic representation of the convergent pathophysiological mechanisms linking Hyperthyroidism and Anxiety Disorders.
Excess thyroid hormones act directly on the central nervous system, increasing beta-adrenergic receptor density and altering neurotransmitter balance. Concurrently, hyperthyroidism stimulates the HPA axis, leading to a chronic stress response. Furthermore, autoimmune thyroid diseases (like Graves' disease) are associated with gut microbiota dysbiosis, leading to systemic inflammation and potential disruption of the blood-brain barrier, further exacerbating psychiatric symptoms.
Abbreviations: 5-HT: Serotonin; HPA: Hypothalamic-Pituitary-Adrenal; HPT: Hypothalamic-Pituitary-Thyroid; NE: Norepinephrine; T3: Triiodothyronine; fT4: Free Thyroxine.
The clinical presentation of both hyperthyroidism and primary anxiety disorders can exhibit significant phenomenological overlap. Shared symptoms include restlessness, irritability, insomnia, fine tremor, palpitations, and impaired concentration [5].
However, specific features help differentiate between the two conditions. Symptoms more characteristic of hyperthyroidism are typically metabolic in nature, including unexplained weight loss despite an increased appetite, heat intolerance, muscle weakness (particularly proximal myopathy), thyroid gland enlargement (goiter), and ocular signs typical of Graves’ disease [18, 30]. Symptoms pointing towards primary anxiety disorders, depending on the specific diagnosis, include chronic, pervasive worry (GAD), unexpected or intense, time-limited episodes of fear (panic attacks), or strong avoidance behaviors related to specific feared situations (phobias) [19]. Unlike HT, primary anxiety disorders are characterized by a chronic course without the defining metabolic symptoms of thyrotoxicosis [19].
The increased prevalence of psychiatric disorders, including anxiety and depression, among patients with hyperthyroidism is consistently affirmed by numerous studies [2, 20]. Other severe psychiatric symptoms that may manifest during the course of hyperthyroidism include agitation, apathy, mania, delusional behavior, hallucinations, acute psychosis, or dementia, with these severe presentations often occurring in older adults [7].
The association between HT and anxiety is complex and manifests in various clinical scenarios, extending beyond active Graves' disease [2, 8, 20, 21]. One critical scenario is subclinical hyperthyroidism induced by L-thyroxine (LT-4) therapy during the treatment of differentiated thyroid carcinoma [8]. In these patients, studies have shown an exacerbation of anxiety symptoms, as measured by the Beck Anxiety Inventory (BAI), and a decline in quality of life (measured by the SF-36 scale) that correlates with fT4 levels [22]. This finding implies the necessity of enhanced clinical caution in LT-4 dosing in this indication to prevent iatrogenic adverse effects [8].
In clinical practice, it is paramount to differentiate whether the psychiatric symptoms are primary or are secondary consequences of the underlying thyroid hormonal imbalance [4, 21]. Consequently, routine assessment of Thyroid-Stimulating Hormone (TSH), free thyroxine (fT4), and T3 concentrations should be mandatory [4]. This comprehensive screening should also be considered for patients with treatment-resistant anxiety and other chronic psychiatric disorders [4].
Furthermore, a prospective cohort study revealed that existing anxiety symptoms are significantly associated with an increased risk of developing hyperthyroidism [21]. This risk escalates with symptom severity, tripling in men experiencing high levels of anxiety [21]. While this trend is less pronounced in women, possibly due to physiological and psychological gender differences in stress coping mechanisms, it underscores the need for active screening for thyroid function abnormalities in patients presenting with new-onset or severe depressive and anxiety symptoms [21]. Despite the recognized high co-occurrence of these conditions, the underlying causal link remains unclear and warrants further investigation [21].
Psychological factors also play a critical role in the course of thyroid diseases. Stressors related to managing a chronic condition, its impact on daily functioning, and social relationships can exacerbate pre-existing psychiatric symptoms associated with hyperthyroidism, leading to a deterioration of mental health and secondarily complicating the overall treatment of the thyroid disease [23]. Clinicians must be cognizant of this vicious cycle mechanism and focus on both the biological and psychological aspects of the disease in patient care [23]. This integrated approach is particularly vital given that this patient group is also at heightened risk for depression [24].
Awareness of the clinical overlap between hyperthyroidism and anxiety disorders is essential for several reasons: i) to ensure correct psychiatric treatment by acknowledging thyroid dysfunction as a modifiable risk factor; ii) to facilitate proactive monitoring of patients with Autoimmune Thyroid Disease (AITD) for an elevated risk of psychiatric disorders; and iii) to avoid excessive levothyroxine dosing in patients with subclinical hypothyroidism, thereby preventing the induction of exogenous hyperthyroidism and associated psychiatric disturbances [8].
To facilitate the clinical differentiation between hyperthyroidism related anxiety manifestations and primary anxiety disorders, the key overlapping symptoms and distinguishing features described in the literature are summarized in Table 1.
Table 1. Clinical Overlap and Differentiating Features Between Hyperthyroidism and Primary Anxiety Disorders.
| Feature Domain | Clinical Symptom/Sign | Hyperthyroidism | Primary Anxiety Disorders | Clinical Note on Overlap/Differentiation |
| Somatic/Physical | Palpitations or Tachycardia | Common; often persistent, even at rest or during sleep. | Common; usually situational or episodic (panic attacks). | High Overlap. Resting pulse rate is a key differentiator. |
| Tremor | Common; typically a fine motor tremor. | Common; often coarser shaking related to acute fear. | High Overlap. The quality of tremor may differ. | |
| Diaphoresis (Sweating) | Common; related to heat intolerance. Skin feels warm/moist. | Common; Often "cold sweats" related to acute sympathetic discharge. | Moderate Overlap. Skin temperature can help differentiate. | |
| Heat Intolerance | Classic sign. | Uncommon. Patients may report hot flashes during panic. | Differentiating Feature. | |
| Weight Change | Weight loss despite increased or normal appetite (metabolic). | Weight loss usually associated with decreased appetite due to stress, or weight gain (comfort eating). | Differentiating Feature. Appetite vs. weight trajectory is crucial. | |
| Ocular Signs | Graves' ophthalmopathy (exophthalmos, lid lag) in GD. | Absent. | Differentiating Feature (specific to Graves'). | |
| Psychological/Cognitive | Anxiety/Worry | Secondary; often described as "internal restlessness" or chemical agitation. | Primary; pervasive, chronic worry (GAD) or catastrophic fear (Panic). | High Overlap. The quality of anxiety often differs (somatic-driven vs. cognitively-driven). |
| Irritability/Agitation | Very Common. | Common. | High Overlap. | |
| Concentration/Memory Impairment | Common ("brain fog"). Often reversible with euthyroidism. | Common; related to pervasive worry and distractibility. | High Overlap. | |
| Sleep Disturbances | Insomnia common due to hypermetabolic state. | Insomnia (difficulty falling/staying asleep) related to worry. | High Overlap. | |
| Pathophysiological Basis | Primary Driver | Excess Thyroid Hormones (T3/fT4) driving beta-adrenergic receptor upregulation. | Dysregulation of neurotransmitters (GABA, 5-HT, NE) and limbic system neural circuits. | Distinct primary mechanisms with converging downstream sympathetic effects. |
Footnotes for Table 1: GAD: Generalized Anxiety Disorder; GD: Graves' Disease; 5-HT: Serotonin; GABA: γ-aminobutyric acid; NE: Norepinephrine; T3: Triiodothyronine; fT4: Free Thyroxine.
Anxiety disorders, which affect approximately 34% of US adults during their lifetime, are strongly associated with severe psychological distress and generalized impairment [3]. In patients with hyperthyroidism, this distress is compounded: excess thyroid hormones compromise emotional stability, resulting in increased irritability and negatively impacting interpersonal relationships, thereby diminishing patient functioning and overall quality of life [4, 25].
The severity of this clinical burden was demonstrated in a naturalistic, prospective study of hyperthyroidism patients where standardized instruments were used for assessment, including GAD-7 (anxiety), PHQ-9 (depression), WSAS (functional impairment), and EuroQol ED5D (quality of life) [2]. A considerable proportion of these patients (40.5%) exhibited moderate to severe anxiety, with an even higher proportion (50.6%) showing moderate to severe depression. Critically, scores for anxiety, depression, and functional impairment were significantly correlated with each other and inversely correlated with quality of life [2]. This high prevalence of co-occurring anxiety has also been confirmed in other studies focusing on patients with Graves’ disease [2,21,26], collectively emphasizing the decisive role of psychiatric comorbidity in the functional decline of these individuals [27].
The complexity of thyroid hormone modulation extends beyond emotional symptoms. A study focusing on patients with subclinical hyperthyroidism, developed during suppressive levothyroxine therapy for differentiated thyroid carcinoma, observed a significant deterioration in executive functions, psychomotor speed, and attention on standard neuropsychological tests [7]. Levothyroxine is one of the most frequently prescribed medications globally, yet it possesses a narrow therapeutic index. Overdosing can induce symptoms of iatrogenic hyperthyroidism, causing secondary psychiatric symptoms and severely reducing patients' quality of life and functional capacity [8]. Therefore, the imperative for accurate dosing and rigorous hormonal monitoring following dose adjustments must be strongly underscored [8].
The significant societal cost stemming from higher rates of long-term sickness absence among patients with thyroid diseases also highlights the complexity of the problem and its impact beyond individual health, affecting the overall functioning of society [28].
Management of hyperthyroidism (HT) is based on the severity of the case and involves pharmacological antithyroid medications, radioactive iodine therapy (RAI), or thyroid surgery. Given that none of these modalities represents an ideal definitive treatment, novel methods to care for patients continue to emerge [9].
The identification of underlying hyperthyroidism as the cause of psychiatric symptoms should not relieve clinicians of the obligation to provide formal psychiatric consultation and management [2, 20]. Successful treatment of hyperthyroidism and the normalization of free thyroxine (fT4) levels typically lead to a significant amelioration of psychiatric symptoms. Nevertheless, a substantial proportion of patients continue to experience psychiatric morbidity despite achieving euthyroidism [2]. Therefore, routine mental health assessment is essential for early diagnosis and effective therapy. Screening patients for psychiatric symptoms and disorders can not only improve disease control but, crucially, enhance their overall quality of life [20]. While primary endocrinological therapy substantially reduces the prevalence of certain symptoms and disorders, it may prove insufficient for others [20, 21]. In these patients experiencing residual or persistent symptoms, the initiation of supplemental treatment with selective serotonin reuptake inhibitors (SSRIs) or serotonin-norepinephrine reuptake inhibitors (SNRIs) is clinically warranted. Furthermore, psychotherapy, particularly Cognitive Behavioral Therapy (CBT), is a crucial intervention, as specific meta-analyses show that it can serve as a first-line therapy for certain anxiety disorders, such as Generalized Anxiety Disorder (GAD) [29]. Consequently, exploring new combined therapeutic strategies, including concurrent antithyroid treatment with psychotropic medications and psychotherapy, remains a vital area of research.
Beta-adrenergic antagonists (beta-blockers) are utilized to mitigate the somatic manifestations of anxiety, such as palpitations, tachycardia, and tremor [30]. It is important to note that, while highly effective in controlling adrenergic symptoms, these agents do not address the underlying etiology of the anxiety. Furthermore, beta-blockers are associated with frequent adverse events (e.g., bradycardia, hypotension, erectile dysfunction) and are contraindicated in specific patient populations, such as those with atrioventricular block or bronchial asthma [30].
There is a notable absence of recent randomized controlled trials evaluating the efficacy and safety of SSRIs and SNRIs for anxiety management during the active thyrotoxic phase. This evidence gap necessitates clinical prudence, often dictating that the decision to initiate these psychotropic agents be postponed until hormonal stability (euthyroidism) is achieved [3, 27]. Similarly, dedicated research into the effectiveness of psychotherapy in treating anxiety disorders within this patient cohort is lacking.
Given that the gut microbiota is known to be altered in autoimmune thyroid diseases (AITD), some preliminary studies propose the use of probiotics as supportive therapy which, when co-administered with antithyroid medications like methimazole, may lead to improved thyroid function in Graves’ disease [9, 27]. Since investigations into the comprehensive relationship between probiotics, hyperthyroidism, and neurotransmitter balance are currently limited, further dedicated research is warranted [30]. This line of inquiry is particularly significant due to existing research on the potential influence of microbiota dysbiosis on the pathogenesis of psychiatric conditions, including anxiety disorders [13]. Addressing these complex biological findings, future therapeutic strategies should concentrate on targeted intervention in the disrupted signaling pathways and hormonal axes implicated in the shared pathophysiology. For instance, some research suggests that anxiety symptoms might be mitigated through BDNF/TrkB signaling modulation [31]. Furthermore, interactions between the hypothalamic-pituitary-thyroid (HPT) and hypothalamic-pituitary-adrenal (HPA) axes should be prioritized for future therapeutic investigation [8, 10]. The gut-thyroid axis interacts significantly with trace elements; for instance, Selenium supplementation has been shown to modulate gut microbiota and reduce thyroid autoantibodies, potentially alleviating psychological distress [32]. Emerging evidence also suggests that short-chain fatty acids (SCFAs) produced by gut bacteria play a crucial role in regulating the blood-brain barrier permeability in Graves' disease [33].
The landscape of research into thyroid-related psychiatric disorders is evolving through the application of precision medicine technologies. Recent large-scale genetic analyses have confirmed shared hereditary predispositions between autoimmune thyroid diseases and psychiatric conditions, identifying significant overlaps in risk loci [34]. Another area of emerging research focuses on the genetic underpinnings connecting endocrine hormones and psychiatric disorders, utilizing advanced genomic mapping to identify shared genetic predispositions [35]. Such investigations can pinpoint specific genetic markers that confer susceptibility to both hyperthyroidism and anxiety disorders, paving the way for personalized therapeutic strategies [36].
Recent studies also indicate that COVID-19 infection can exacerbate anxiety and depression in Graves' disease patients, with symptom count acting as an independent risk factor for thyroid function alterations, highlighting the influence of acute stressors on the endocrine-psychiatric interface. Furthermore, the interplay between endocrine imbalances, immune responses, and the gut-brain axis in the context of viral infections like COVID-19 presents complex challenges for both treatment and prognosis in hyperthyroid patients [37].
The exploration of novel therapeutic approaches has also expanded to include the modulation of the gut microbiome, with recent findings suggesting that probiotic supplementation, particularly with Bifidobacterium longum, can enhance the efficacy of conventional antithyroid treatments and influence the gut-thyroid axis in Graves' disease [38,39]. This suggests a critical intersection between microbial interventions and endocrine regulation, potentially offering a new avenue for ameliorating both thyroid dysfunction and associated psychiatric comorbidities [40, 41].
Future research should also explore how specific endocrine-related hormones, such as TRH, and antibodies, including TPOAb and TRAb, might serve as biomarkers for predicting the onset or severity of anxiety in hyperthyroid patients [42].
Comprehensive understanding of these complex interrelationships, integrating insights from neuroendocrinology, immunology, and gut microbiota research, is crucial for developing novel, multi-modal interventions that address the root causes of endocrine-driven anxiety [43,44].
The co-occurrence of hyperthyroidism and anxiety disorders represents a clinically relevant and multidimensional problem that cannot be adequately explained by endocrine or psychiatric frameworks alone. The literature analyzed in this review consistently indicates that normalization of thyroid hormone levels, while essential, does not invariably lead to complete resolution of anxiety symptoms. Several observational and prospective studies report persistent psychological complaints after restoration of euthyroidism, suggesting that anxiety manifestations may not be solely a direct and reversible consequence of hormonal excess but may involve additional mechanisms. These observations are reported in clinical and population based studies assessing quality of life and mental health outcomes in patients treated for hyperthyroidism and Graves disease.
From a pathophysiological perspective, acute anxiety symptoms during thyrotoxicosis can be plausibly explained by the direct effects of excess thyroid hormones on central adrenergic activity and neurotransmitter balance, as described in mechanistic and clinical reviews. However, the persistence of anxiety symptoms after biochemical normalization points toward more complex and potentially longer lasting processes. Several included sources discuss the possible contribution of neuroinflammation, immune mediated mechanisms, and alterations along the gut brain axis, particularly in autoimmune thyroid disease. Experimental and translational studies provide supportive but indirect evidence for these pathways, while clinical confirmation remains limited.
The reviewed literature also reveals a gap between endocrine and psychiatric clinical practice. Endocrinology oriented guidelines primarily focus on biochemical targets such as TSH and free thyroid hormone levels, whereas structured assessment of mental health outcomes is often limited or absent. Conversely, psychiatric literature addressing anxiety disorders frequently considers thyroid dysfunction only as a differential diagnosis based on basic thyroid function tests, without systematic evaluation of autoimmune markers. This separation is highlighted in reviews and cohort studies and may contribute to delayed recognition of comorbid conditions and incomplete management of affected patients.
Therapeutic considerations discussed in the literature further reflect these limitations. Beta adrenergic blockers are consistently described as effective for controlling somatic symptoms of anxiety during active hyperthyroidism, yet they do not address central psychological components. Evidence regarding the use of antidepressant or anxiolytic pharmacotherapy in patients with active or recently treated thyrotoxicosis is sparse. The absence of randomized controlled trials specifically addressing the safety and efficacy of SSRIs or SNRIs in this context forces clinicians to extrapolate from general anxiety treatment guidelines, despite known alterations in metabolism and receptor sensitivity associated with thyroid hormone excess.
Several limitations of the available literature must be acknowledged. A substantial proportion of the included studies are observational or retrospective in design, which limits causal inference between thyroid dysfunction and specific anxiety phenotypes. Definitions of anxiety disorders and assessment tools vary widely across studies, ranging from formal diagnostic criteria to symptom based questionnaires, reducing comparability. Similarly, the characterization of hyperthyroidism differs between sources, with inconsistent differentiation between overt, subclinical, and treatment induced states. Experimental and preclinical studies provide mechanistic insights but cannot be directly translated into clinical recommendations.
In summary, the discussion of current evidence underscores the need for more integrated and methodologically robust research. Future studies should prioritize longitudinal designs that combine hormonal, immunological, and psychometric assessments to clarify temporal relationships and therapeutic implications. Until such data are available, management of patients with coexisting hyperthyroidism and anxiety disorders must rely on individualized, multidisciplinary approaches informed by the heterogeneous evidence base summarized in this review.
This narrative review provides an integrated synthesis of current evidence on the relationship between hyperthyroidism and anxiety disorders, focusing on shared pathophysiological mechanisms, clinical symptom overlap, diagnostic complexity, and consequences for quality of life. The analyzed literature confirms that hyperthyroidism represents a clinically significant and often underrecognized contributor to anxiety related symptomatology, warranting systematic consideration in patients presenting with psychiatric complaints.
The findings demonstrate that normalization of thyroid hormone levels, although a fundamental therapeutic objective, does not reliably result in complete resolution of anxiety symptoms or functional impairment. This underscores the multifactorial nature of anxiety manifestations in hyperthyroid patients and indicates that a purely endocrine centered treatment strategy is frequently insufficient. Effective management therefore requires coordinated interdisciplinary collaboration between endocrinology and psychiatry to improve diagnostic accuracy and clinical outcomes.
At the mechanistic level, the reviewed data support the involvement of convergent neuroendocrine and immunological pathways, including altered neurotransmission, interactions between the HPT and HPA axes, autoimmune processes, and emerging gut brain axis mechanisms. However, the available evidence remains heterogeneous and is largely derived from observational and translational studies, limiting causal inference.
Future research should prioritize longitudinal and interventional designs integrating hormonal, immunological, and psychometric measures. Particular attention should be directed toward understanding the persistence of anxiety symptoms after restoration of euthyroidism and toward evaluating targeted psychotherapeutic and pharmacological interventions in hyperthyroid populations. Addressing these gaps is essential for developing evidence based, multidisciplinary strategies aimed at improving long term functional outcomes and quality of life in affected patients.
Conceptualization: Szymon Zysiak, Julia Wawerska, Łukasz Deska methodology:Dawid Głaz, Maksymilian Głaz, Natalia Kamińska, Jędrzej Zaguła, Cezary Kosmecki
Software: Cezary Kosmecki, Maksymilian Głaz
Investigation: Szymon Zysiak, Łukasz Deska
Resources: Wojciech Sołtys, Magdalena Stolarczyk, Jędrzej Zaguła
Data curation: Łukasz Deska, Wojciech Sołtys, Magdalena Stolarczyk, Natalia Kamińska Writing - rough preparation: Cezary Kosmecki, Jędrzej Zaguła
Writing - review and editing: Dawid Głaz, Maksymilian Głaz, Wojciech Sołtys
Visualization: Natalia Kamińska, Julia Wawerska
Supervision: Szymon Zysiak, Julia Wawerska, Łukasz Deska
Project administration: Szymon Zysiak
All authors have read and approved the final version of the manuscript.
Artificial intelligence tools were not used for data collection, data analysis, or interpretation of the results presented in this article and were applied only for language editing and stylistic refinement of the manuscript text.