- Home
- About the Journal
- Peer Review
- Editorial Board
- For Authors
- Reviewer Recognition
- Archive
- Contact
- Impressum
- EWG e.V.
Cite as: Archiv EuroMedica. 2026. 16; 1. DOI 10.35630/2026/16/Iss.1.007
Obesity is a chronic disease associated with increased cardiometabolic risk and requires long term multidisciplinary management. Semaglutide, a glucagon like peptide 1 receptor agonist, has emerged as an effective pharmacological option for weight management. The aim of this narrative review is to analyze current clinical evidence on the efficacy, safety, and adverse event profile of semaglutide in adults with overweight or obesity.
A narrative review of publications indexed in PubMed, Scopus, Web of Science, Embase, Google Scholar, and ResearchGate was conducted. Eligible studies published between 2020 and 2026 included randomized controlled trials, cohort studies, systematic reviews, meta analyses, and international clinical guidelines. Only studies involving adult participants meeting established indications for pharmacological obesity treatment, defined as a body mass index of at least 30 kg per square meter or at least 27 kg per square meter with weight related comorbidities, were included.
The review synthesizes evidence from the STEP 1 to 8 clinical trials evaluating once weekly subcutaneous semaglutide in diverse clinical and ethnic populations. Across all trials, semaglutide in combination with lifestyle modification was consistently associated with clinically meaningful weight reduction compared with placebo. Comparative data demonstrate greater weight loss with semaglutide than with liraglutide in adults without diabetes mellitus. Semaglutide was effective in Asian populations characterized by increased cardiometabolic risk at lower body mass index values and was associated with reductions in visceral adipose tissue. In individuals with type 2 diabetes mellitus, weight loss was less pronounced but remained clinically relevant. The most frequently reported adverse events were gastrointestinal in nature and were predominantly mild to moderate in severity, with low rates of serious adverse events and treatment discontinuation.
Current clinical evidence indicates that once weekly subcutaneous semaglutide, used as an adjunct to lifestyle intervention, is an effective and generally well tolerated pharmacological option for weight management in adults with overweight or obesity. Maintenance of weight reduction requires continued treatment, supporting the concept of obesity as a chronic condition requiring long term therapeutic strategies.
Keywords: semaglutide; obesity; overweight; GLP 1 receptor agonist; weight management.
Characterized by the pathological accumulation of adipose tissue, obesity is a chronic disorder that compromises physiological well-being and correlates with higher rates of secondary illnesses and fatality [1,21,23]. The World Health Organization (WHO) generally classifies adult obesity as having a Body Mass Index (BMI) of 30 kg/m² or higher. Nevertheless, relying solely on BMI fails to account for the diverse nature of the condition, particularly regarding an individual's specific body fat distribution, associated illnesses, and overall clinical risk profile [2,22]. Over the past few decades, the global prevalence of obesity has escalated, driven by enhanced food security, socioeconomic advancements, and significant shifts in lifestyle patterns. These changes, fueled by globalization and the industrialization of food production, have fostered obesogenic environments. Consequently, obesity has emerged as a major public health emergency, affecting over one billion individuals as rates continue to climb internationally [3]. In 2021, elevated Body Mass Index (BMI) was responsible for approximately 3.7 million fatalities linked to noncommunicable diseases (NCDs). These included conditions such as cardiovascular and chronic respiratory diseases, diabetes, various malignancies, and neurological or digestive disorders [4,23,34,42].
A holistic, evidence-based approach to obesity management integrates behavioral therapy, nutritional guidance, and physical activity with pharmacological or surgical interventions, tailored to the specific needs of the patient [5,26].
GLP-1 (Glucagon-like peptide-1), a primary incretin hormone synthesized by intestinal L cells, modulates glycemic control by stimulating insulin secretion and suppressing glucagon release in a glucose-dependent manner. Its weight-management properties are primarily mediated through the arcuate nucleus of the hypothalamus, where it facilitates appetite suppression and decreased caloric intake by amplifying satiety signals and attenuating hunger cues [6,27,28,30,37,38].
Semaglutide, which functions as a GLP-1 receptor agonist, is indicated for the management of type 2 diabetes through either weekly subcutaneous injections (0.25, 0.5, and 1 mg) or daily oral administration (3, 7, and 14 mg). Furthermore, since June 2021, the FDA has authorized the use of semaglutide for chronic weight control, specifically utilizing higher weekly injectable doses of 1,7 and 2,4 mg [7,22,31,33,35,38,42,43].
This narrative review offers a distinctive contribution to the field by providing a unified analysis of the entire STEP 1–8 trial sequence. While most existing literature addresses these trials separately, our synthesis explores the drug’s multidimensional impact across varying ethnic and clinical cohorts. By integrating long-term maintenance data with results from intensive lifestyle interventions, we provide a novel evidence-based perspective on semaglutide’s role in modern obesity management, highlighting its benefits beyond mere weight reduction.
The objective of this review is to analyze current evidence of the efficacy, safety and adverse events of semaglutide in adults with overweight or obesity.
Research objectives of the article:
This article is a narrative review based on publications from PubMed, Scopus and Web of Science. Publications from January 2020 to January 2026 were considered. The search terms used were: semaglutide AND obesity OR side effects OR adverse effects OR efficacy.
Inclusion criteria were: randomized controlled trials, cohort studies, systematic reviews, meta-analyses, and international clinical guidelines reporting on treatment of obesity with semaglutide. Only studies enrolling adult patients who fulfilled the eligibility criteria for pharmacological intervention—namely, a BMI greater than 30 kg/m² or at least 27 kg/m² in the presence of one or more weight-related comorbidities—were considered for inclusion.
Exclusion criteria were: case reports, conference abstracts, non-peerreviewed articles, and publications without clear data on interventions or without relevance to semaglutide.
The initial database search yielded 221 records. After removal of duplicate entries and application of the inclusion and exclusion criteria, 44 publications were retained for qualitative synthesis. The selection process was based on relevance to the objectives of the review and the availability of extractable clinical data related to efficacy and safety outcomes.
As this work represents a narrative review, no formal protocol registration, standardized risk of bias assessment, or independent duplicate screening procedures were applied. The methodology is intended to provide a structured and transparent overview of the available clinical evidence rather than a quantitative synthesis.
A series of STEP (Semaglutide Treatment Effect in People with obesity) clinical trials were designed to test the efficacy and safety of semaglutide 2.4 mg in various clinical scenarios.[36] STEP 1 studied if semaglutide has a significant impact on weight loss in the general population of people without diabetes. Enrolling criteria were adults who did not have diabetes, BMI of 30 kg/m² and higher or BMI of 27 kg/m² and higher with at least weight-related coexisting conditions. The study group was divided into 2 subgroups of semaglutide 2.4 mg intake weekly and visually matching placebo. All participants had to undergo counselling sessions, set a daily calorie deficit of 500 kcal in diet and increase time of physical activity. After 68 weeks semaglutide led to approximately -14.9% body weight loss over up to -2.4% in the placebo group, improved blood pressure, lipid levels, and decreased CRP. Not only did the mean body weight decrease but more participants who were in the semaglutide group achieved weight reduction of clinical importance (5% or more of weight loss) [8,43]. STEP 2 included people with BMI ≥27 kg/m² and type 2 diabetes mellitus. It was carried out to determine if semaglutide is also effective in case of people with disturbed sugar metabolism. Achieved weight loss was lower than in STEP 1 (-9.6% with 2.4 mg semaglutide vs placebo -3.4%). Similarly improvements were noted in blood pressure, lipid levels and decreased CRP and also fasting glucose levels and glycated hemoglobin HbA1c decreased. This study also included a third group which was given an intermediate dose of 1 mg semaglutide. It achieved slightly less body weight - 6.9% than 2.4 mg dose but still remains statistically relevant. Laboratory tests results of 1 mg semaglutide were between 2.4 mg dose and placebo. On the other hand adverse effects were less frequent in smaller doses [9,43]. STEP 3 modified STEP 1 approach with radical lifestyle intervention. Participants followed a low-calorie diet (1000–1200 kcal/d) for 8 weeks, followed by a hypocaloric diet (1500–1800 kcal/d) for the subsequent 60 weeks, along with intensive behavioral therapy. Physical activity was prescribed for them. Starting from 100 minutes increased systematically up to 200 minutes (spread across 4-5 days). The mean change in body weight from baseline was –16.0% for semaglutide 2.4 mg, compared to –5.7% for placebo [10]. The durability of the weight loss effect was studied in STEP 4. After 20 weeks of run-in period of increasing semaglutide dose to 2.4 mg one of the study groups was switched to placebo. People on semaglutide continued to lose weight contrary to the placebo group which gradually regained weight. Similar effects of worsening of clinical markers were observed in systolic pressure, fasting glucose levels, HbA1c, lipid profile and even mental health [11]. STEP 5 studied the efficacy of treatment over a very long time of 104 weeks. While most of the trials lasted 68 weeks, this particular study was twice as long. It proved the weight loss effect can be sustained for a full 2 years and the drug's safety profile remains stable. A mean weight loss of -15.2% at the end of 104 weeks was achieved. It has a similar effect to STEP 1 -14.9% after 68 weeks. Differences with placebo groups are -12.6% for STEP 5 and -12.4% for STEP 1 [12]. Standard BMI ranges do not fully reflect the metabolic risk in Asians. Obesity and related disorders, such as insulin resistance, develop at much lower body mass index values for this race. This is due to the specific distribution of adipose tissue, where metabolically active visceral fat accumulates at earlier stages of overweight than in other populations [29]. For this reason, STEP 6 focused on the Japanese and South Korean populations. Once again semaglutide in two doses 2.4 and 1.7 mg (plus lifestyle modification) achieved mean bodyweight change -13.2% and -9.6% respectively, by week 68. The authors performed an additional CT scan on the subgroup. A significant reduction in abdominal visceral fat was demonstrated, which directly translates into improved metabolic parameters and reduction in cardiovascular risk. Significant visceral fat reductions (≥20%) were observed in 84% and 62% of participants treated with semaglutide 2.4 mg and 1.7 mg respectively, whereas only 21% of the placebo group reached this threshold [13]. STEP 7 investigated the same problem as the previous issue's study. It was designed with the population of mostly China and other Asian countries. Patients received standard treatment with semaglutide 2.4 mg or placebo in addition to diet and exercise. The study duration was 44 weeks. It was shorter than the standard 68 weeks in STEP 1. Nevertheless, patients taking semaglutide lost an average of 12.1% of their body weight, while the placebo group lost only 3.6%. As previously, significant improvements were noted in waist circumference, systolic blood pressure, and fasting glucose and lipid levels [14]. STEP 8 compared semaglutide with previous-generation substance. The authors confronted semaglutide 2.4 mg weekly with 3.0 mg liraglutide daily. Participants with obesity and overweight were included. It did not allow participants with diabetes to take part. By the end of 68 weeks, results of body weight change were -15.8% for semaglutide vs. -6.4% for liraglutide. Also semaglutide 2.4 mg showed superior efficacy over liraglutide 3.0 mg across a broad range of cardiometabolic markers. Specifically, semaglutide led to significantly greater reductions in absolute body weight, waist circumference, and C-reactive protein levels. Furthermore, significant improvements were observed in fasting plasma glucose, HbA1c, and lipid profiles, including total cholesterol and triglycerides. Semaglutide also demonstrated a lower permanent discontinuation rate at 13.5%, significantly outperforming liraglutide percentage of 27.6% [15]. The clinical efficacy outcomes of the STEP 1 to 8 trials are summarized in Table 1.
Table 1. Summary of clinical efficacy outcomes in STEP clinical trials
| Study | Population | Diabetes status | Duration weeks | Semaglutide dose mg | Comparator | Mean body weight change percent | Key additional outcomes |
| STEP 1 | Adults with overweight or obesity | No diabetes | 68 | 2.4 | Placebo | decrease 14.9 | Improvements in cardiometabolic parameters |
| STEP 2 | Adults with overweight or obesity | Type 2 diabetes | 68 | 2.4 | Placebo | decrease 9.6 | Reduced HbA1c and fasting glucose |
| STEP 3 | Adults with overweight or obesity with intensive lifestyle intervention | No diabetes | 68 | 2.4 | Placebo | decrease 16.0 | Enhanced weight loss under behavioral therapy |
| STEP 4 | Adults with overweight or obesity after run in period | No diabetes | 68 | 2.4 | Placebo | weight maintenance with continuation | Prevention of weight regain |
| STEP 5 | Adults with overweight or obesity | No diabetes | 104 | 2.4 | Placebo | decrease 15.2 | Sustained efficacy over two years |
| STEP 6 | East Asian adults with overweight or obesity | With or without diabetes | 68 | 2.4 and 1.7 | Placebo | decrease 13.2 at 2.4 mg and decrease 9.6 at 1.7 mg | Reduction of visceral adipose tissue |
| STEP 7 | Adults with overweight or obesity from China | No diabetes | 44 | 2.4 | Placebo | decrease 12.1 | Improvements in waist circumference and lipid profile |
| STEP 8 | Adults with overweight or obesity | No diabetes | 68 | 2.4 | Liraglutide 3.0 | decrease 15.8 versus decrease 6.4 | Greater efficacy compared with liraglutide |
Semaglutide exhibited a favorable safety and tolerability profile throughout the analyzed clinical trials. Although a high percentage of participants (87.2%) in STEP 1-8 reported at least one adverse event (AE), these rates were remarkably similar to those observed in the placebo group (88.3% vs 85%, respectively) The most common gastrointestinal adverse events observed in the semaglutide group compared to placebo were diarrhea, nausea, constipation and vomiting as detailed in Table 1 [8-15, 35]. The majority of these occurrences were of mild to moderate intensity and typically resulted in full recovery or resolution. These adverse events were predominantly reported during the dose-escalation period, as semaglutide was being increased to the maintenance level [8,11,12,13,22,24,30,32,33]. Moreover the incidence of adverse events—mostly gastrointestinal—was similar across the semaglutide and liraglutide arms in STEP 8, mirroring findings from other clinical trials [15,19,20]. Discontinuation rates resulting from adverse events were approximately 4.2% across the study, a figure that includes participants from the placebo cohort. A higher rate of treatment discontinuation due to adverse events was observed in the semaglutide cohort (5.3%) compared to the placebo group (3%), with gastrointestinal complications being the primary cause [8-15]. Permanent treatment discontinuation rates due to adverse events were comparable between the semaglutide 2.4 mg (5/171; 3%) and 1.7 mg (3/82; 3%) cohorts in STEP 6, with no notable differences observed [13,25]. While in STEP 2 treatment discontinuation were reported in 6.2% (25/353) in the 2.4 mg semaglutide group vs 5% (5/171) in the 1.0 mg semaglutide group [9]. Semaglutide also demonstrated a lower permanent discontinuation rate at 13.5%, significantly outperforming liraglutide, which had a rate of 27.6% (with discontinuation due to adverse events at 3.2% vs. 12.6%, respectively) [15]. Serious adverse events (SAEs) occurred in 5.3% of the semaglutide cohort and 3.0% of the placebo group [8-15]. The proportion of participants in the semaglutide group who reported hepatobiliary disorders (mainly cholelithiasis) was greater than in the placebo group [8,10,12]. The observed frequency of hepatobiliary disorders may be partially driven by rapid weight reduction, which is a well-established risk factor for the formation of gallstones [16,17,18]. A total of eight deaths were reported throughout the studies; however, the independent external event adjudication committee determined that none were related to the trial product [8-15].
The data on adverse effects acquired during the STEP 1-8 studies have been collated and are presented in the Table 2 below.
Table 2. Summary table of adverse effects reported during STEP 1-8 clinical trials
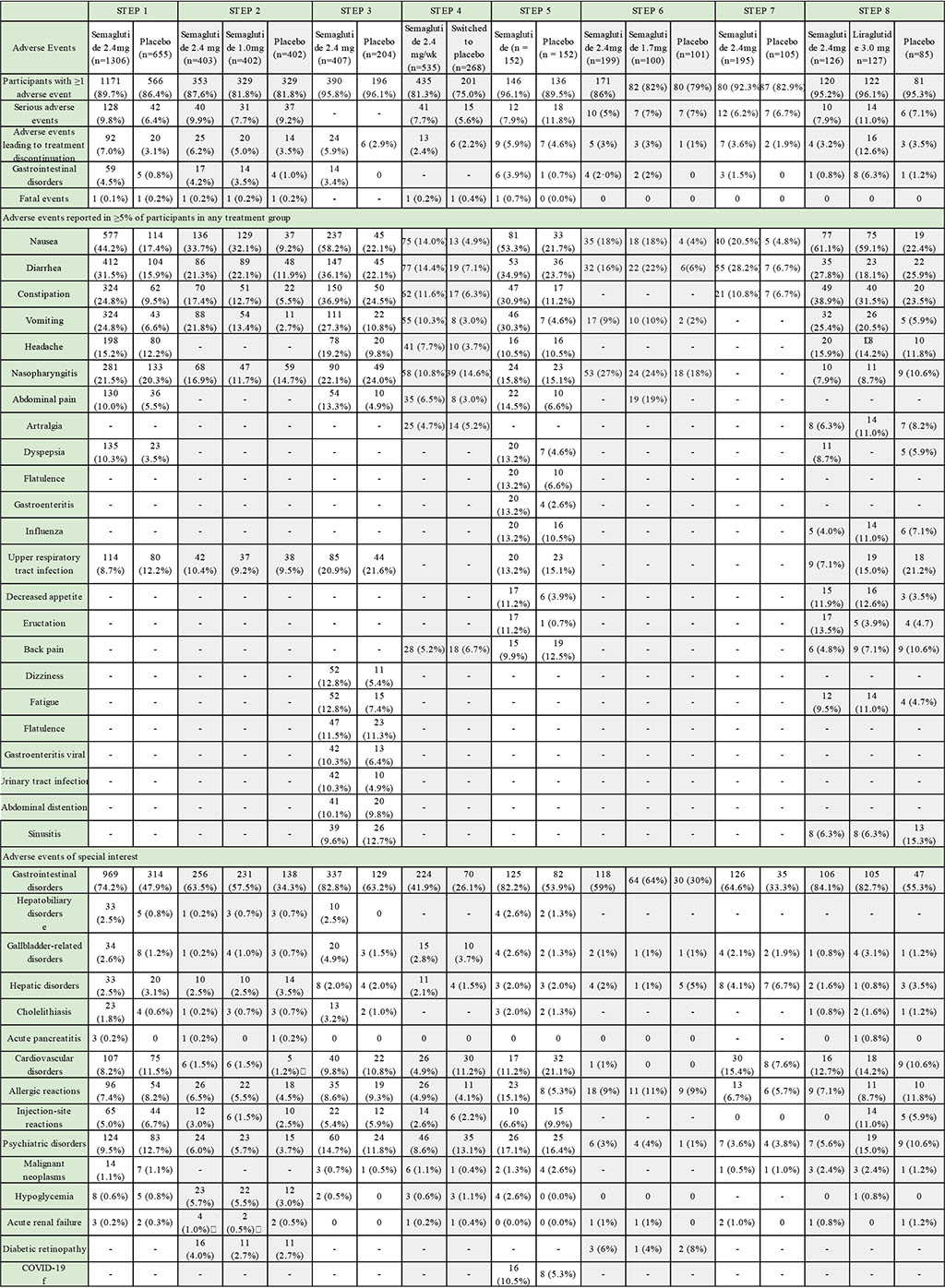
Summary tables of adverse effects from each study were consolidated into a single unified table. To ensure clarity, isolated adverse reactions representing negligible percentages were excluded from this analysis.
This narrative review summarizes the results of the STEP 1 to 8 clinical trials evaluating the use of semaglutide in adults with overweight or obesity [8,9,10,11,12,13,14,15]. The presented data demonstrate a consistent and sustained body weight reduction associated with semaglutide 2.4 mg in combination with lifestyle modification across all analyzed clinical scenarios. Weight loss was observed both in individuals without disturbances of glucose metabolism and in patients with type 2 diabetes mellitus, although the magnitude of the effect was lower in the diabetic population [9].
Comparisons with placebo across all STEP trials confirm the superiority of semaglutide in terms of weight reduction and improvement of cardiometabolic parameters, including blood pressure, lipid profile, and inflammatory markers [8,10,11,12]. The results of the STEP 8 trial indicate greater efficacy of semaglutide compared with liraglutide, accompanied by a comparable safety profile and a lower rate of permanent treatment discontinuation [15].
Data derived from Asian populations in the STEP 6 and STEP 7 trials are of particular relevance. These studies demonstrate that semaglutide is effective in populations characterized by a higher cardiometabolic risk at lower body mass index values [13,14,29]. Additional analysis of visceral adipose tissue in STEP 6 showed a clinically meaningful reduction in abdominal fat, which is directly associated with decreased metabolic and cardiovascular risk [2,13].
The safety profile of semaglutide summarized in this review is characterized by a high incidence of adverse events, predominantly gastrointestinal in nature, consistent with previously published data [17,22]. Most adverse events were mild to moderate and occurred primarily during the dose escalation phase [21,22]. The observed increase in hepatobiliary events aligns with the well documented association between rapid weight loss and the risk of gallstone formation [16,17].
Findings from STEP 4 and STEP 5 highlight the necessity of continued semaglutide administration to maintain achieved weight loss, reflecting the chronic nature of obesity as a disease [11,12,34]. Overall, the available evidence supports the role of semaglutide as an effective component of long term pharmacological obesity management within a multidisciplinary treatment framework [5,23,26].
This review has limitations inherent to its narrative design. A formal systematic review protocol was not applied, no prior methodological registration was performed, and standardized tools for risk of bias assessment of the included studies were not used.
A quantitative meta analysis was not conducted, limiting the ability to formally assess heterogeneity across studies or to calculate pooled effect estimates. The analysis is based on published clinical trial data, and the potential impact of publication bias cannot be excluded.
In addition, the review predominantly includes data from randomized controlled STEP trials, while evidence from real world clinical practice is limited [33,43]. This may restrict the generalizability of the findings to routine clinical settings and specific patient subgroups.
These limitations should be taken into account when interpreting the conclusions of this review.
This narrative review systematizes clinical evidence from the STEP 1 to 8 trials regarding the efficacy and safety of semaglutide in adults with overweight or obesity. The analyzed data demonstrate that once weekly subcutaneous semaglutide, used in combination with lifestyle modification, is consistently associated with clinically meaningful weight reduction compared with placebo across a range of clinical settings and patient populations.
The review confirms that semaglutide treatment is characterized by a predictable safety profile, with adverse events predominantly involving the gastrointestinal tract and most frequently occurring during the dose escalation phase. The majority of these events are mild to moderate in severity, while rates of serious adverse events and permanent treatment discontinuation remain low.
Comparative analysis shows that semaglutide provides greater weight reduction than liraglutide when both agents are administered as adjuncts to lifestyle counseling in adults without diabetes mellitus. Evidence from studies conducted in Asian populations indicates that semaglutide is effective in clinical contexts associated with increased cardiometabolic risk at lower body mass index values and is associated with reductions in visceral adipose tissue.
In patients with impaired glucose metabolism or type 2 diabetes mellitus, semaglutide remains effective, although the magnitude of weight loss is reduced compared with non diabetic populations, while clinically relevant metabolic benefits are preserved. Long term data indicate that maintenance of weight reduction requires continued pharmacological therapy, underscoring the chronic nature of obesity.
Overall, the findings of this review support the role of semaglutide as an effective pharmacological component of multidisciplinary obesity management, addressing the predefined objectives related to efficacy, safety, comparative effectiveness, and population specific responses.
Conceptualization: Zuzanna Gąsior, Przemysław Krukowski
Methodology: Małgorzata Landowska, Iga Poprawa
Formal analysis: Michał Maciejowski, Marta Kwiatkowska
Investigation: Mateusz Zdaniewicz, Michał Stermach
Writing-rough preparation: Zuzanna Gąsior
Writing-review and editing: Przemysław Krukowski
Supervision: Nina Kubikowska, Patryk Krawczyk
All authors have read and agreed with the published version of the manuscript.
The authors declare that no artificial intelligence tools were used in the generation, writing, editing, or revision of this manuscript. All content was created solely by the authors.
The article did not receive any funding.
Authors declare no conflicts of interest.