- Home
- About the Journal
- Peer Review
- Editorial Board
- For Authors
- Reviewer Recognition
- Archive
- Contact
- Impressum
- EWG e.V.
Cite as: Archiv EuroMedica. 2026. 16; 2. DOI 10.35630/2026/16/Iss.2.12
Eosinophilic esophagitis is a chronic immune mediated disease characterized by eosinophilic infiltration of the esophageal mucosa and symptoms of esophageal dysfunction. Despite the use of proton pump inhibitors, topical corticosteroids, and dietary interventions, a substantial proportion of patients does not achieve adequate clinical response. Biologic therapy has emerged as a targeted treatment option, however available evidence remains heterogeneous and requires critical synthesis.
To provide a comprehensive overview of current evidence on biologic therapy in eosinophilic esophagitis and to evaluate its clinical relevance based on histological, endoscopic, and symptomatic outcomes.
This study was designed as a narrative review. Literature was identified using PubMed and complementary open access sources, including PubMed Central, Google Scholar, DOAJ, ClinicalTrials.gov, and other open repositories. The search covered publications up to March 16, 2026, with prioritization of studies from the last five years. Selection of sources was based on relevance to the objectives of the review rather than systematic screening. A total of 54 publications were included.
Biologic therapies consistently reduce eosinophilic inflammation, however their clinical efficacy varies depending on the targeted pathway. Dupilumab demonstrates the most consistent improvement across histological, endoscopic, and clinical outcomes. In contrast, therapies targeting IL 5, IL 5R, and IL 13 are associated with reduction in eosinophil counts without consistent symptom improvement. Emerging therapies targeting TSLP, Siglec 8, IL 15, and KIT demonstrate biological activity, but their clinical significance remains uncertain.
Biologic therapy in eosinophilic esophagitis shows heterogeneous effectiveness and depends on the targeted mechanism. Dupilumab currently represents the most clinically validated option. Other biologic agents demonstrate partial or preliminary effects. The findings should be interpreted with caution due to the narrative design of the review and non-systematic study selection.
Keywords: eosinophilic esophagitis, EoE, biologic therapy, dupilumab, benralizumab, mepolizumab, reslizumab, cendakimab
Eosinophilic esophagitis is a chronic inflammatory disease with increasing prevalence and a risk of progressive esophageal remodeling. In recent years, there has been a rapid rise of the incidence and prevalence of EoE [1,2]. The clinical presentation differs depending on age.[3] Adolescents and adults most commonly experience dysphagia and food bolus impaction, while younger children tend to present with non-specific symptoms such as abdominal pain, vomiting, and failure to thrive; infants may also have feeding difficulties.
Although no endoscopic findings are pathognomonic, features such as longitudinal furrows, esophageal rings, mucosal pallor, white plaques, strictures, and a narrow-caliber esophagus may suggest EoE. [4]
Histological features that could be found in patients with EoE are eosinophilic microabscesses, basal cell hyperplasia, elongation of rete-peg, fibrosis of the subepithelial lamina propria, extracellular eosinophil granules, superficial layering of eosinophils and increases in other cell types, such as lymphocytes [5]
EoE predominantly affects males [6–8] and is more common in Caucasians than in other ethnic groups, as well as in adults compared to children and the elderly. [9] Allergic conditions, especially IgE-mediated food allergies, significantly increase the risk. [9,10]. Early-life factors such as antibiotic use [11], NICU stay [12], cesarean section [11] , maternal fever [13], prematurity [13], and PPI use are associated with higher risk, possibly through microbiome disruption and impaired immune tolerance [11]. EoE was also associated with the decreasing prevalence of Helicobacter pylori [14], although the mechanism of this correlation remains unknown. In contrast, breastfeeding [12] and early exposure to furry pets may be protective [13].
Various genetic and environmental factors, as well as individual immune characteristics, play a role in the pathogenesis of EoE [3,15,16], which was schematically presented in figure 1. Genetic predisposition is associated with single nucleotide polymorphisms in the genes encoding immune-related molecules, such as thymic stromal lymphopoietin (TSLP), eotaxin-3 (CCL-26), calpain-14 (CAPN14) [17–20]. As indicated in figure 1, in genetically predisposed individuals, food allergens, and less commonly inhaled allergens, as well as infectious agents, and irritants cause the esophageal epithelium to produce eotaxin-3 (CCL-26), interleukin 25 (IL-25), interleukin 33 (IL-33), and thymic stromal lymphopoietin (TSLP)[21]. Eotaxin-3 is a main eosinophil-selective chemotactic factor that recruits eosinophils from blood and stimulates them to infiltrate esophageal mucosa. Its activity is enhanced by IL-4, IL-5, IL-13. In patients with EoE, the level of CCL-26 in the esophageal epithelium and serum is elevated during active inflammation [20,22]. As shown in figure 1, TSLP, IL-15, IL-25, and IL-33 promote the generation of Th2 cells and the activation of epithelial cells, dendritic cells, NK cells, mast cells, basophils, and iLC2 [23,24]. This leads to the secretion of cytokines characteristic of the Th2-dependent immune response, such as IL-4, IL-5, IL-13 and eosinophils stimulation [23]. Eosinophils secrete cytotoxic proteins and inflammatory mediators such as eosinophil peroxidase, eosinophil cationic protein, and major binding protein, resulting in tissue damage and esophageal motility disorders due to M2 muscarinic receptor dysfunction. Mast cells amplify inflammation through mediators (histamine, PGD2, leukotrienes) [25]. Interleukine-13, by inducing calpain-14 with protease activity, causes a decrease of desmoglein-1, filaggrin, zona occludens-1 and claudin-1. These changes prompt reduction of intercellular connections, impaired epithelial barrier function and tissue remodeling [23,26]. The main profibrotic cytokine TGF-β released by mast cells and other inflammatory cells, together with IL-1β, TNF-α, and IL-4, play a major role in esophageal fibrosis. They stimulate the transformation of fibroblasts into myofibroblasts, the synthesis of fibronectin, type I collagen, periostin, and smooth muscle actin [27] After contraction, myofibroblasts secrete extracellular matrix components, including type 1 collagen, to stabilize their new, contracted position, leading to progressive narrowing and dysmotility of the esophagus [28], as reflected in Figure 1.
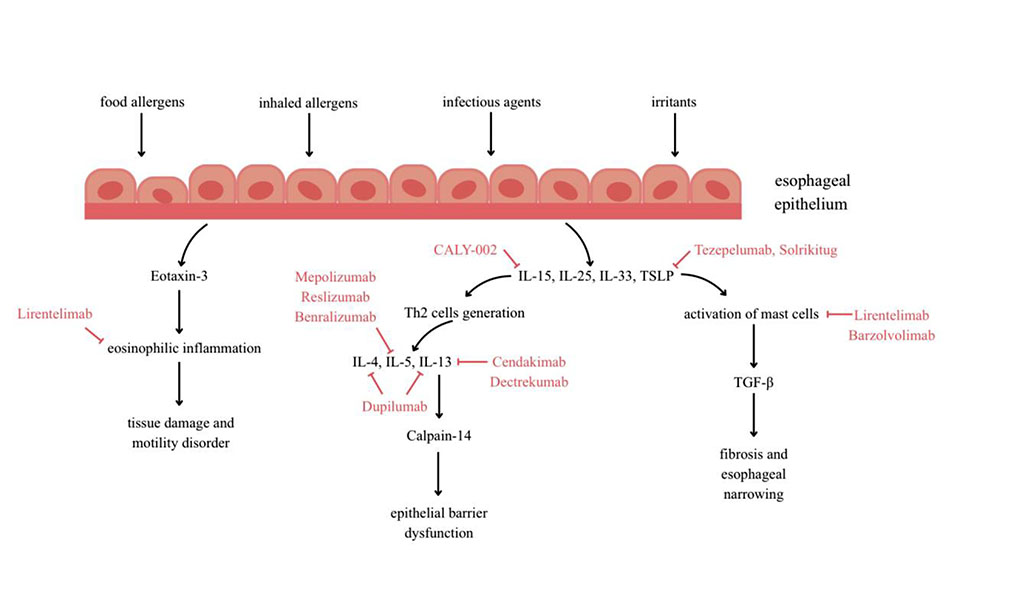
Figure 1 (Image by Author) illustrates the pathogenesis of eosinophilic esophagitis, with particular emphasis on the molecular targets of biologic therapies
A substantial proportion of patients do not achieve adequate response to standard therapy, including proton pump inhibitors, topical corticosteroids, and dietary interventions. In this context, biologic therapy is considered a promising targeted treatment approach. Biologic agents are considered when the conventional therapy fails to achieve or maintain remission, cause intolerable side effects, or when long-term steroid use is a concern. However, the available evidence remains heterogeneous, and the relationship between histological response and clinical outcomes is not clearly established, which necessitates its systematization.
This study provides a synthesis of current data on biologic therapy in eosinophilic esophagitis with a focus on different therapeutic targets, including IL 4, IL 13, IL 5, TSLP, Siglec 8, IL 15, and KIT. Particular attention is given to the discrepancy between histological improvement and clinical response, as well as to the role of tissue remodeling and mast cells in the persistence of symptoms. The study systematizes emerging therapeutic approaches and outlines their limitations.
Aim of the study: to analyze current evidence on biologic therapy in eosinophilic esophagitis, with a focus on mechanisms of action and clinical effectiveness of the agents.
Research objectives:
This study was designed as a narrative review with a structured literature search.
A comprehensive search of the literature was conducted using the following freely accessible databases and sources: PubMed, PubMed Central, Google Scholar, DOAJ, ClinicalTrials.gov, as well as open access aggregators including BASE, CORE, and Semantic Scholar. The search covered publications from January 1, 2021 to March 16, 2026. Earlier studies were included selectively when they provided essential data on pathogenesis or biologic therapy relevant to current clinical practice.
The search strategy was structured using Boolean operators. The main search query included combinations of the following terms: “eosinophilic esophagitis” AND “biologic therapy” OR “dupilumab” OR “mepolizumab” OR “benralizumab” OR “reslizumab” OR “cendakimab” OR “tezepelumab” OR “lirentelimab” OR “siglec-8” OR “IL-13” OR “IL-5” OR “TSLP”. Additional terms such as “pathogenesis”, “clinical trial”, and “treatment outcomes” were used to refine the search where appropriate.
The selection process included initial screening of titles and abstracts, followed by full text assessment for eligibility. The final inclusion of studies was based on their relevance to the objectives of this narrative review and the availability of clinically meaningful data. A total of 85 sources were included in the final analysis. The selection process was descriptive and did not follow a formal systematic review protocol.
Biologic drugs are a promising and rapidly developing therapeutic option for eosinophilic esophagitis, especially in patients with severe or refractory disease. Dupilumab (anti-IL4-R monoclonal antibody) is currently the only registered drug for the treatment of EoE. Other biological drugs remain in the research phase.The characteristics of the most important biological drugs used or studied in the treatment of eosinophilic esophagitis, including their mechanism of action and clinical trial results, are presented below.
Dupilumab is a fully human monoclonal antibody. It binds to the interleukin-4 receptor alpha subunit (IL‐4Rα) shared by both IL‐4 and IL‐13 receptors. By blocking these receptors, dupilumab suppresses IL-4 and IL-13 signaling, which leads to the alleviation of type 2 inflammation caused mainly by this cytokine pathway [29]. In published phase 3 randomized controlled trials (RCT) including pediatric patients aged ≥ 1 year and adults dupilumab, compared to placebo, significantly increased the percentage of patients achieving histologic remission, defined as a peak esophageal intraepithelial eosinophil count of ≤6 per high-power field (HPF), improved endoscopic image as assessed by the EoE Endoscopic Reference Score (EREFS), and reduced the frequency and severity of dysphagia assessed using standardized symptom questionnaires such as Dysphagia Symptom Questionnaire (DSQ) and Pediatric Eosinophilic Esophagitis Sign/Symptom Questionnaire - Caregiver version (PESQ-C)[30–32]. After treatment, esophageal biopsy also reveals ablation of basal cell hyperplasia and elimination of dilated intracellular spaces. In addition, dupilumab has a beneficial effect on the biomechanical properties of the esophageal wall, increasing its distensibility and potentially fractionally reversing remodeling changes. To date, dupilumab is the only biologic drug that simultaneously improves histological, endoscopic, and clinical outcomes, while demonstrating a favorable long-term safety profile , with the most common adverse reaction being reactions at the site of administration [33]. Food and Drug Administration (FDA) approved medication for the treatment of EoE in May 2022, with expansion in use to patients as young as 1 year of age weighing at least 15 kg in 2024. Standard therapy consists of subcutaneous administration once a week for 24 weeks [29].
Cendakimab (formerly known as RPC4046 or CC-93538) and Dectrekumab (QAX576) are monoclonal antibodies targeting IL-13 with a similar mechanism of action to dupilumab. In a double-blind, placebo-controlled phase II trial, cendakimab significantly reduced peak eosinophil counts, endoscopic severity, and EoE-related symptoms (as measured by the Dysphagia Symptom Diary, DSD) compared to patients receiving placebo [34]. In phase 3 trials cendakimab showed statistically significant reduction of dysphagia and improvement in histological and endoscopic outcomes which were sustained through 48 weeks. Although adverse effects were relatively common, the safety profile was acceptable and did not limit the use of the therapy [30]. Dectrekumab reduced esophagus eosinophilia and improved the expression of genes associated with eosinophil chemotaxis (eotaxin-3), tissue remodeling (periostin), epithelial barrier integrity (desmoglein 1), and mastocytosis, but complete histologic remission was not achieved.
Despite this, a tendency toward improvement in clinical symptoms, especially the severity of dysphagia, was observed [35].
Mepolizumab and reslizumab are humanized monoclonal antibodies with the same mechanism of action. They bind circulating IL-5, thereby preventing it from binding to the interleukin-5 receptor (IL-5R). As described earlier, IL-5 plays a key role in the maturation, activation, trafficking and survival of eosinophils. Both mepolizumab and reslizumab significantly reduce the number of eosinophils in peripheral blood and limit their infiltration into the esophageal mucosa [36,37]. After treatment with mepolizumab, reduced expression of TGF-β, a key marker associated with fibrosis and esophageal remodeling, was observed in esophageal tissues, which was not confirmed after reslizumab therapy [38]. In phase II RCT studies, mepolizumab decreased the number of eosinophils in the mucosa and reduced the severity of endoscopic inflammatory changes such as edema, exudates and furrows according to the EREFS scale. However, patients randomly assigned to the mepolizumab group did not show significant improvement in dysphagia assessed through the EoE Symptom Activity Index (EEsAI) in comparison to patients randomly assigned to the placebo group. A lack of response was also observed in the case of other histological features, such as basal zone hyperplasia, spongiosis, and lamina propria fibrosis. Importantly, these results were observed in a population of patients with EoE that can be classified as severe.[36]. In studies conducted in the pediatric population, a reduction in esophageal eosinophilia and improvement in endoscopic findings were observed; however, as in adults, no relevant reduction in clinical symptoms was demonstrated after mepolizumab therapy [39]. Reslizumab was tested in a study involving children and adolescents with EoE. Similar to mepolizumab, the therapy resulted in a significant decrease in esophageal eosinophils; nevertheless, the therapy did not lead to the expected improvement in symptoms [40].On the other hand, during 9 years of follow-up with reslizumab treatment, all patients showed no dysphagia or strictures on endoscopic evaluation despite a relatively unrestricted diet [41].
Another biological drug being studied for the treatment of EoE is benralizumab. It is an anti–interleukin-5 receptor α monoclonal antibody that induces direct, rapid, and near-complete eosinophil depletion through antibody-dependent cellular cytotoxicity (ADCC). In the latest phase 3 trial benralizumab substantially reduced the number of eosinophils in the esophagus mucosa, but did not result in a greater reduction in dysphagia symptoms in comparison to placebo. No apparent improvement in endoscopic findings or histologic remission was observed [42].
TSLP is a relatively new and promising target for biological therapy of EoE. As an epithelial cytokine initiating the type 2 inflammatory response, TSLP participates in the earliest stages of pathogenesis even before the activation of classic Th2 mediators. Tezepelumab and solrikitug are monoclonal antibodies against TSLP. Randomized, double-blind, placebo-controlled trials are currently underway to evaluate the efficacy of tezepelumab and solrikitug administered subcutaneously in patients with EoE. A single clinical report indicates that tezepelumab may contribute to EoE remission and reverse characteristic molecular changes, providing strong justification for further research into this therapeutic strategy [43]. Another case report suggested that tezepelumab may lead to meaningful clinical improvement in patients with severe, treatment-refractory EoE. In this case, a patient who did not respond to PPIs, FTCs , FED, or anti-IL-5–targeted biological treatment (mepolizumab and benralizumab) showed improvement in symptoms, endoscopic findings, and histologic features after initiation of tezepelumab, supporting the need of further clinical research on this therapeutic approach. [44]
Sialic-acid-binding immunoglobulin-like lectin-8 (Siglec-8) is an inhibitory receptor selectively expressed on human eosinophils and mast cells. It has been observed that its activation by monoclonal antibodies - lirentelimab (AK002) - induces eosinophil depletion through ADCC and inhibits IgE-dependent mast cells activation. In a phase 2/3 study involving adolescents and adults, lirentelimab significantly reduced eosinophil count but did not produce an apparent improvement in symptoms as assessed by the DSQ scale, although a trend toward improvement in DSQ scores was observed in adolescents compared with placebo [45]. Research on lirentelimab is still ongoing to more accurately determine its efficacy and potential benefits in different patient groups.
IL-15 contributes to the pathogenesis of EoE by promoting the activation of cytotoxic T lymphocytes, NK cells, and ILC2s, thereby amplifying inflammatory responses.
A phase 1a/1b RCT study evaluated single ascending doses of anti-IL-15 monoclonal antibody (CALY-002) in healthy volunteers and repeated dosing in patients with EoE. Preliminary clinical data indicate encouraging efficacy, with marked decreases in esophageal eosinophilic inflammation, alongside improvement in dysphagia. [46]
The KIT receptor is essential in the survival and activation of mast cells, positioning it as a potential attractive target for novel biologic interventions [47]. This was examined in the second phase of a randomized, double-blind, placebo controlled trial evaluating the safety and efficacy of barzolvolimab, an anty-KIT monoclonal antibody. Despite profound depletion of mast cells, no clinical improvement was observed.[48]
Biological therapies in EoE demonstrate a consistent ability to reduce eosinophilic inflammation, however their clinical efficacy remains variable depending on the targeted pathway. As summarized in Table 1, only selected agents translate anti inflammatory effects into consistent clinical benefit, while others show discordance between histological and symptomatic outcomes.
Dupilumab remains the only biologic that consistently improves histological, endoscopic and clinical outcomes simultaneously, as reflected in Table 1, supporting the central role of the IL 4 and IL 13 axis in EoE pathogenesis [31,32,49]. Across randomized trials, histological remission rates with dupilumab exceed those observed with placebo by a clinically meaningful margin, with parallel improvements in symptom scores and endoscopic findings. The maintenance of efficacy regardless of prior corticosteroid exposure further highlights its potential utility in both treatment naive and refractory populations [50,51]. In addition, data indicate that dupilumab may allow reintroduction of previously identified trigger foods without worsening disease activity [52].
In contrast, therapies targeting IL 5 or IL 5R such as mepolizumab, reslizumab and benralizumab, although associated with marked reductions in eosinophil counts, do not translate into consistent symptom improvement. As shown in Table 1, the reduction in tissue eosinophilia is not accompanied by comparable changes in dysphagia scores or patient reported outcomes. This dissociation between histological and clinical response suggests that reduction of eosinophils alone is insufficient to control disease activity. This is further supported by the lack of improvement in structural features such as basal zone hyperplasia and fibrosis [36–42]. Similarly, IL 13 targeted therapies demonstrate beneficial effects on histological and endoscopic endpoints, with measurable reductions in inflammatory markers, while their impact on symptoms remains less consistent, indicating limited reversibility of established tissue remodeling [30,34,35].
Evidence on IL 15 directed intervention is currently scarce, and available data summarized in Table 1 do not allow quantitative assessment of clinical efficacy.
Emerging therapies targeting upstream mediators such as TSLP may provide broader modulation of the inflammatory cascade. As indicated in Table 1, these approaches act at earlier stages of disease development, which may be relevant given the complexity of type 2 inflammation [43]. Lirentelimab, targeting Siglec 8, affects both eosinophils and mast cells and represents a broader immunomodulatory strategy. However, clinical effects remain modest, with only limited reductions in symptom scores despite measurable biological activity. This suggests that the degree of mast cell modulation achieved is insufficient to significantly impact symptom generation.
These findings, together with the limited clinical efficacy observed in IL 5 targeted therapies, support the concept that mechanisms beyond eosinophil depletion contribute to disease activity. Mast cells are increasingly recognized as key contributors to inflammation and tissue remodeling, and their role may explain the persistence of symptoms despite reduction of eosinophilic infiltration.
Targeting the KIT receptor, which is essential for mast cell survival and activation, may therefore represent a more direct therapeutic strategy. As shown in Table 1, anti KIT therapies such as barzolvolimab are currently under investigation. Available early phase data indicate biological activity, however robust quantitative clinical outcomes are not yet established. Table 1 summarizes key characteristics of investigated biologic drugs.
Table 1. Key characteristics of the aforementioned biologic drugs
| Drug | Study type | Population | Dose and duration | Histological effect | Clinical symptoms | Endoscopic finding | Key conclusions | Reference |
| Dupilumab | RCT phase 3, registered by FDA (2022) | Adults + pediatric ≥1 year | 300 mg sc.weekly 24 weeks | Significant remission | improvement | improvement | Only registered biologic improving histology, symptoms, and endoscopy simultaneously | [32,49] |
| Cendakimab | RCT phase 3 | 12 to 75 years of age | 360 mg sc. weekly 48 weeks | significant remission | improvement | improvement | Significant clinical, histological and endoscopic improvement with a favorable safety profile | [30] |
| Dectrekumab | RCT phase 2 | 18-50 years of age | (6 mg/kg) sc.at weeks 0, 4, and 8 for 6 months. | reduced esophageal eosinophilia, improved expression of EoE-relevant esophageal transcripts, no full remission | trend only | no data | Significant reduction of of intraepithelial esophageal eosinophil counts and improvement of dysregulated esophageal disease-related gene expression | [35] |
| Mepolizumab | RCT phase 2 | 16-75 years of age | 300 mg sc.monthly for 3 months | reduced esophageal eosinophilia, no full remission | no improvement | improvement | no improvement of dysphagia vs placebo, reduction of eosinophil count and improvement of endoscopic finding at 3 months, however no additional benefit was found long term | [36] |
| Reslizumab | RCT phase 2 + 9-year follow-up | children and adolescents | iv. infusions of 1, 2, or 3 mg/kg at weeks 0, 4, 8, and 12. | reduced esophageal eosinophilia, no full remission | Partial improvement in follow-up | no data | not correlated with changes in eosinophil count improvement of symptoms, which was observed in both patient groups | [40] |
| Benralizumab | RCT phase 3 | 12-65 years of age | 30mg sc. every 4 weeks for 24 weeks | reduced esophageal eosinophilia, no full remission | no improvement | no improvement | histologic response in more patients in the benralizumab group vs placebo, however no reduction of dysphagia was observed vs placebo | [42] |
| Tezepelumab | RCT phase 3 ongoing | 12-80 years of age | sc., 52 weeks | no data | no data | no data | no data | [53] |
| Solrikitug | RCT phase 2, ongoing | adults | sc., 24 weeks | no data | no data | no data | no data | [54] |
| Lirentelimab | RCT, phase 2/3 | ≥12 years of age | high dose (1 mg/kg x 1 dose then 3 mg/kg x 5 doses low dose (1 mg/kg, ) for 6 monthly infusions | reduced esophageal eosinophilia, no full remission | no significant improvement, trend in adolescents | no data | histologic outcomes improvement with no symptom improvement | [45] |
| Barzolvolimab | RCT phase 2 | ≥18years of age | 300 mg sc. weekly for 28 weeks | reduced intrepithelial and cutaneous mast cells count | no improvement | no data | Mast cell depletion, which did not result in clinical improvement | [48] |
Overall, the available evidence suggests that biologic therapies in eosinophilic esophagitis differ substantially in their clinical relevance depending on the targeted pathway. Dupilumab currently represents the most clinically validated option, demonstrating consistent improvement across histological, endoscopic, and symptomatic outcomes. In contrast, therapies targeting IL 5 or IL 5R, as well as some IL 13 directed approaches, appear to provide primarily biological effects with less consistent clinical benefit. Emerging targets, including TSLP, Siglec 8, IL 15, and KIT, remain investigational, and the current evidence should be considered preliminary.
From a clinical perspective, biologic therapy should be interpreted in the context of established treatment strategies. Proton pump inhibitors, topical corticosteroids, and dietary interventions remain first line approaches, while biologic agents may be considered in patients with refractory disease or insufficient response to conventional therapy. However, direct comparative data are limited, and the precise positioning of biologic therapy within treatment algorithms requires further clarification.
This study has limitations inherent to its narrative design. The literature selection was not based on a formal systematic protocol, and the inclusion of studies was guided by relevance rather than predefined quantitative criteria. This approach may introduce selection bias and influence the overall interpretation of the evidence.
Eosinophilic esophagitis remains a clinically relevant disease with persistent inflammation and risk of progressive remodeling, particularly in patients with inadequate response to conventional therapy.
Biologic therapy targets key immunological pathways. Dupilumab demonstrates the most consistent improvement across histological, endoscopic, and clinical outcomes and currently represents the only biologic with reproducible clinical benefit in practice.
Therapies targeting IL 5, IL 5R, and partly IL 13 reduce eosinophilic inflammation, but this does not consistently translate into symptom improvement, indicating that eosinophil reduction alone is insufficient to control disease activity.
Emerging targets such as TSLP, Siglec 8, IL 15, and KIT show biological activity, but clinical evidence remains limited and preliminary.
Overall, the effectiveness of biologic therapy in eosinophilic esophagitis depends on the targeted pathway and ranges from consistent clinical benefit to isolated biological effects without clear symptomatic improvement.
Conceptualization and methodology: Filip Chodań
Investigation and data collection: Filip Chodań, Olaf Helbig, Gabriela Ciszek
Formal analysis: Filip Chodań, Olaf Helbig
Writing - original draft preparation: Filip Chodań, Olaf Helbig, Gabriela Ciszek
Writing - review and editing: Filip Chodań, Olaf Helbig
Supervision - Filip Chodań
All authors read and approved the final version of the manuscript and agree to be
accountable for all aspects of the work.
Artificial intelligence tools, such as ChatGPT and other OpenAI systems, were used to support language refinement, structural improvement, and the development of certain text sections (including results and conclusions). All AI-generated contributions were thoroughly reviewed and verified by the authors.